 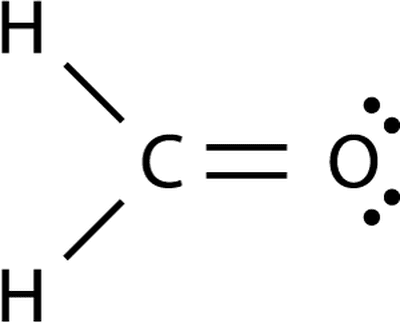
As we can see, the dots around the atoms represent the number of valence electrons. In this molecule, carbon will be the central atom, and the rest(hydrogen) atoms will be the surrounding ones. Now we have to identify the atom that has to be placed in the center, so the central atom is the one that has the least electronegativity. So total number of valence electrons in the molecule will be 4+4 = 8. 
The number of valence electrons in carbon are 4 and in hydrogen, there is one ( as there are four hydrogen atoms in the molecule, the number of valence electrons will be 4). So the number of valence electrons in methane: According to the formula there is one carbon and 4 hydrogens in the structure. We must know the number of valence electrons in order to draw the structure. So we are going to understand how to draw hydrogen and carbon lewis dot structure step by step. By using the concept of lewis dot structure for representing methane we can understand the concept of bonding in the molecule better. The simplest compound that is formed from hydrogen and carbon lewis dot structure is methane. We shall understand this better by studying various carbon Lewis dot structures. The reason carbon can form bonds with various elements is due to properties like catenation, tetra valency, average electronegativity, etc. In this article we are going to study various carbon Lewis dot structures in detail.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
June 2023
Categories |
 RSS Feed
RSS Feed
